
Zhixin Zhang, Yehong Wang, Jianmin Lu, Min Wang, Jian Zhang, Xuebin Liu, Feng Wang*
Inorganics, 2017, 5(4), 75
We herein report the synthesis of 3-methyl-1,3-butanediol from isobutene and HCHO in water via a Prins condensation-hydrolysis reaction over CeO2, which is a water-tolerant Lewis acid catalyst. The CeO2 exhibits significant catalytic activity for the reaction, giving 95% HCHO conversion and 84% 3-methyl-1,3-butanediol selectivity at 150 °C for 4 h. The crystal planes of CeO2 have a signific...
Zhitong Zhao, Yong Liu, Feng Wang, Xuekuan Li, Shuping Deng, Jie Xu*, Wei Wei, Feng Wang*
J. Clean. Prod., 2017, 163, 285-292
Life cycle primary energy demand (PED) and greenhouse gas (GHG) emissions for the production of propylene in China (year 2013) have been carried out by considering several propylene production routes into account, such as catalytic cracking (CC), stream cracking (SC), coal-to-olefins (CTO) and coal-to-propylene (CTP), which cover the entire life cycle including: extraction and transportation...
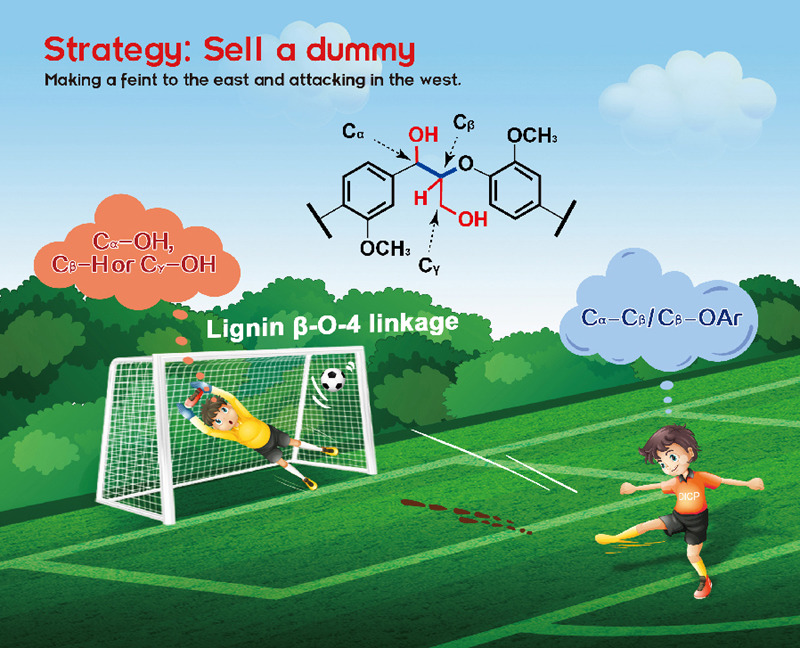
Chaofeng Zhang, Feng Wang*
Chin. J. Catal., 2017. 38(7), 1102-1107
This perspective highlights recent progress on lignin β-O-4 conversion, which focuses on the combinatorial strategies leading to lignin β-O-4 cleavage beginning with adjacent fmunctional group modification, and provides insight into the challenges unexplored in this area.
Tingting Hou, Chaofeng Zhang, Yehong Wang, Zhenting Liu, Zhixin Zhang, Feng Wang*
Catal. Commun., 2017, 94(5), 56-59
A metal-free and efficient approach to N-arylpyrrolidines from arylamines and cyclic ethers catalyzed by hydrogen iodine is described. In this protocol, no additive is added and a wide range of N-arylpyrrolidines are synthesized with up to 99% yield. Reaction mechanism involving iodine radical is proposed.
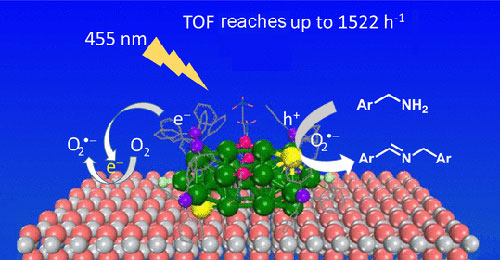
Haijun Chen, Chao Liu, Min Wang, Chaofeng Zhang, Nengchao Luo, Yehong Wang, Hadi Abroshan, Gao Li*, Feng Wang*
ACS Catal., 2017, 7(5), 3632–3638
This work demonstrates the synthesis of an efficient photocatalyst, Au25(PPh3)10Cl2(SC3H6SiO3)5/TiO2, for selective oxidation of amines to imines. The photocatalyst is prepared via hydrolysis of Au25(PPh3)10Cl2[(SC3H6Si(OC2H5)3]5 nanoclusters in the presence of TiO2 support. The gold nanoclusters exhibit good photocatalytic activity using visible light and under mild thermal conditions for the selective oxidation with molecular oxygen (O2). The turnover frequency (TOF) of 4-methylbenzylamine oxi
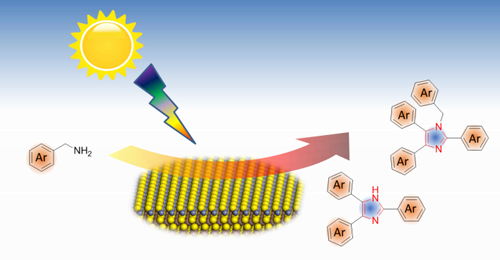
Min Wang, Lihua Li, Jianmin Lu, Nengchao Luo, Xiaochen Zhang, Feng Wang*
Green Chem., 2017, 19(21), 5172-5177
Substituted imidazoles are traditionally synthesized by co-condensation of multiple feedstocks. Herein, we report a new route for the synthesis of substituted imidazoles via photocyclization of readily available amines at room temperature. The reaction is achieved by the visible-light-induced C–C/C–N bond coupling and subsequent dehydrogenation reaction over Mo–ZnIn2S4 as a heterogeneous photocatalyst. A wide range of amines were converted into the corresponding tri- and tetra-substituted imidaz
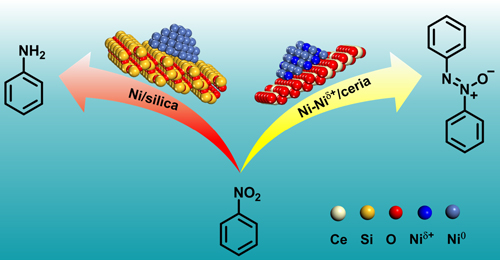
Tingting Hou, Yehong Wang, Jian Zhang, Mingrun Li, Jianmin Lu, Marc Heggen, Carsten Sievers, Feng Wang*
J. Catal., 2017, 353, 107-115
In many catalytic processes, metastable reaction intermediates are more valuable and desirable than final products. Here, we report Ni–Niδ+ clusters on ceria where the extent of reduction of nickel oxide/ceria in H2 has been optimized. This catalyst shows high selectivity in reducing nitrobenzene to azoxybenzene, the latter usually being metastable. Due to strong electronic metal–support interactions between Ni and ceria, mixed Ni–Niδ+ clusters are formed on ceria even after reduction at 500 °C
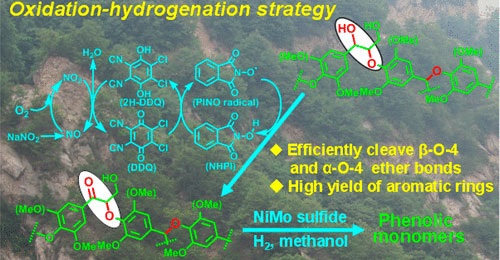
Chaofeng Zhang, Hongji Li, Jianmin Lu, Xiaochen Zhang, Katherine E MacArthur, Marc Heggen, Feng Wang*
ACS Catal., 2017, 7(5), 3419–3429
For lignin valorization, simultaneously achieving the efficient cleavage of ether bonds and restraining the condensation of the formed fragments represents a challenge thus far. Herein, we report a two-step oxidation–hydrogenation strategy to achieve this goal. In the oxidation step, the O2/NaNO2/DDQ/NHPI system selectively oxidizes CαH–OH to Cα═O within the β-O-4 structure. In the subsequent hydrogenation step, the α-O-4 and the preoxidized β-O-4 structures are further hydrogenated over a NiMo
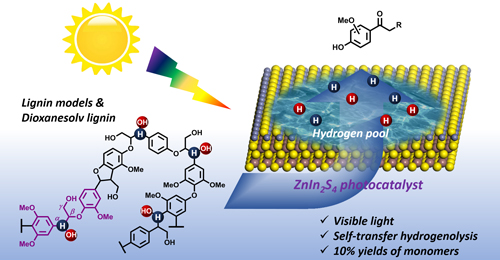
Nengchao Luo, Min Wang, Hongji Li, Jian Zhang, Tingting Hou, Haijun Chen, Xiaochen Zhang, Jianmin Lu, Feng Wang*
ACS Catal., 2017, 7(7), 4571-4580
Obtaining high selectivity of aromatic monomers from renewable lignin has been extensively pursued but is still unsuccessful, hampered by the need to efficiently cleave C–O/C–C bonds and inhibit lignin proliferation reactions. Herein, we report a transfer hydrogenolysis protocol using a heterogeneous ZnIn2S4 catalyst driven by visible light. In this process, alcoholic groups (CαH–OH) of lignin act as hydrogen donors. Proliferation of phenolic products to dark substances is suppressed under visib
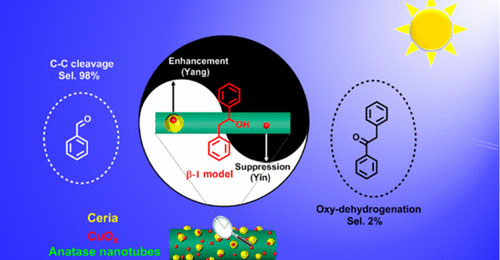
Tingting Hou, Nengchao Luo, Hongji Li, Marc Heggen, Jianmin Lu, Yehong Wang, Feng Wang*
ACS Catal., 2017, 7(6), 3850–3859
Selective cleavage of C–C bonds is pursued as a useful chemical transformation method in biomass utilization. Herein, we report a hybrid CuOx/ceria/anatase nanotube catalyst in the selective oxidation of C–C bonds under visible light irradiation. Using the lignin β-1 model as a substrate offers 96% yields of benzaldehydes. Characterization results by high angle annular dark field scanning transmission electron microscopy (HAADF-STEM) and energy-dispersive X-ray spectroscopy element (EDX) mapping
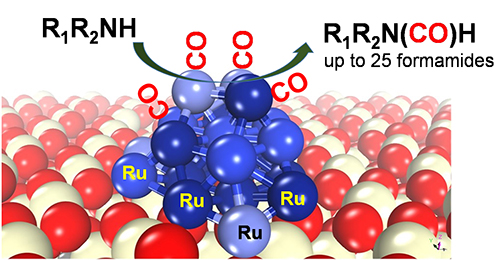
Yehong Wang, Jian Zhang, Haijun Chen, Zhixin Zhang, Chaofeng Zhang, Mingrun Li, Feng Wang*
Green Chem., 2017,19(1), 88-92
We herein report a new strategy of directly converting amines and CO to formamides with 100% atom utilization efficiency. It is suitable for up to 25 amine substrates with no additives. Ru/ceria is found to be an excellent catalyst for this reaction due the efficient co-activation of CO and amine on Ru species.
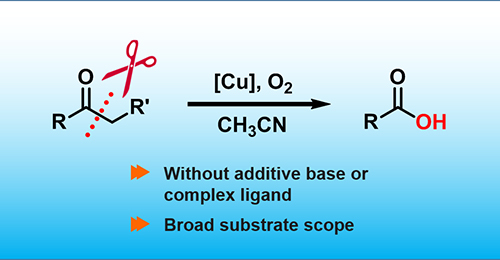
Huifang Liu, Min Wang, Hongji Li, Nengchao Luo, Shutao Xu, Feng Wang*
J. Catal., 2017, 346, 170-179
Catalytic oxidation of C-C bond is a key technology to transform petroleum-based as well as sustainable biomass feedstock into more valuable oxygenates. We herein describe a convenient and useful oxidation strategy of converting ketones into carboxylic acids using homogeneous copper catalyst without additives and with O2 as the terminal oxidant. A wide range of aryl and aliphatic ketones as well as β–O–4 lignin models were selectively oxidized to acids via C-C bond cleavage. Mechanism studies by
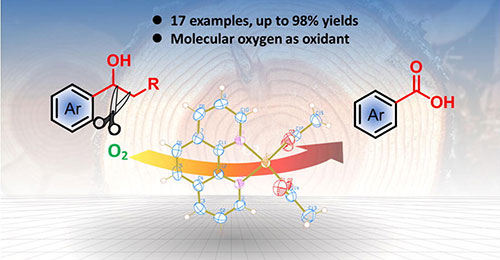
Min Wang, Jianmin Lu, Lihua Li, Hongji Li, Huifang Liu, Feng Wang*
J. Catal., 2017, 348, 160-167
Selective oxidative cleavage of C-C bond is pivotal for producing functionalized molecules, useful for organic synthesis and biomass utilization. We herein report the oxidative C(OH)-C bond cleavage of secondary alcohols to acids over a copper/1, 10-phenanthroline complex with molecular oxygen as the oxidant. A wide range of secondary alcohols are converted into acids with up to 98% yields. More interestingly, it is effective for breaking up lignin model systems into acids, which is rarely achie
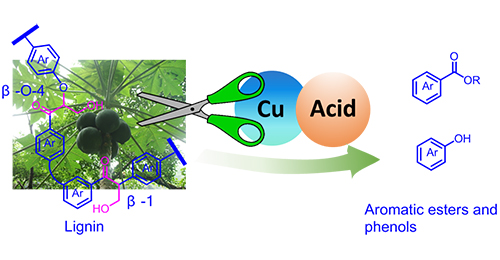
Min Wang, Lihua Li, Jianmin Lu, Hongji Li, Xiaochen Zhang, Huifang Liu, Nengchao Luo, Feng Wang*
Green Chem., 2017, 19(3), 702-706
Depolymerisation of lignin to aromatics is a challenging task. We herein report that a Cu(OAc)2/BF3·OEt2 catalyst is effective in simultaneously cleaving C–C bonds in β-1 and β-O-4 ketones, yielding esters and phenols. In-depth studies show that C–H bond activation is the rate determining step for C–C bond cleavage. BF3·OEt2 promotes the reaction via activating the β-C–H bond. This study offers the potential to obtain aromatic esters from lignin.